TECHNOLOGY
iQ - Integrated Quanticate®
Optimizing the Organizational Performance of the Clinical Trial Process.
Collaboration on documents and distribution of information across a clinical development program for a new product has never been so easy. iQ brings together data, information, knowledge and documents from disparate systems, presented in easy-to-use interfaces, with timely reminders and updates. Through this common platform, you can find the information you need, and get real-time access to the data and knowledge needed to enable faster decisions and optimal commercialization.
Intelligence Portal
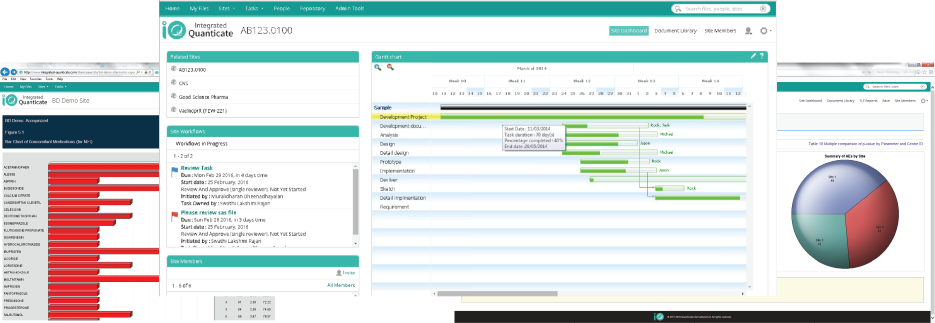
The iQ portal brings together the data, information, and knowledge generated in the development lifecycle of the clinical trial process. The dashboards and visualizations provide fast access to key data and information, from pre-defined and bespoke reports, helping you find meaning and knowledge from your disparate raw data.
Collaboration and Workflow
Document development and review within a version-controlled environment provides a lean approach to your clinical trial process. Workflows model business and regulatory processes, delivering controlled approvals and reviews with timely reminders.
Dashboard and Visualizations
The iQ portal provides trial progress, quality and cost reports. Detailed graphical representation can then be drilled down and filtered when the user requires to enable fast decisions.
Tables, Listings and Figures
iQ offers a standard suite of Tables, Listings and Figures (TLFs) at no additional cost. These near-real time reports permit customers to review the progress of their study in advance of database close, allowing project teams to take a step back from the data and view the trial as a whole. These standard TLFs cover most major interests across study areas; additional custom reports are available for a small charge. Reports include; Demographics, Medical History, Prior Medications, Concomitant Medications, Adverse Events and Vital Signs.
System Features
Using a business intelligence tool to manage the information process flow provides you with reliable access, traceable processes, and single trial or pooled product data to deliver fast answers through:
- Easy to view data from multiple sources in a standardized format
- Full study documentation management and storage including version control
- Task notification for study deliverables and document reviews/approves
- Customized notifications via portal or email
- Shared calendars and discussion forums for a global collaboration
- All content delivered via the portal are available on tablet, PC desktop, and laptop
- Automatic Metadata generation to classify content
- Customizable tags to categorize and search different documents, including both structured and folksonomy tagging
- Global search functionality that reads all documents types including PDFs and all common Office documents
- Gantt Chart views for of project plans, task tracking and resource allocation on your study
Security and Control
- Logon access control across all related systems
- Automated audit administration and easy reporting
- 21CFR Part11 compliance
- Document development and review controls
- Check-in and check-out functionality
- Shared access rules with the data and content store
- Controlled access based on role or group
Request a walkthrough of the iQ Portal
CONTACT DETAILS
Address - UK (HQ):
9-11 Bancroft Court
Hitchin, Hertfordshire
SG5 1LH
United Kingdom
Tel: +44 (0)1462 440 084
Fax: +44 (0)1462 440 086
Email: enquiries@quanticate.com
Address - US:
8601 Six Forks Rd
Suite 400
Raleigh, NC, 27615
United States
Tel: +1-919-882-2016
Email: info@quanticate.com
QUANTICATE
BLOG TOPICS
© 2024 Quanticate