
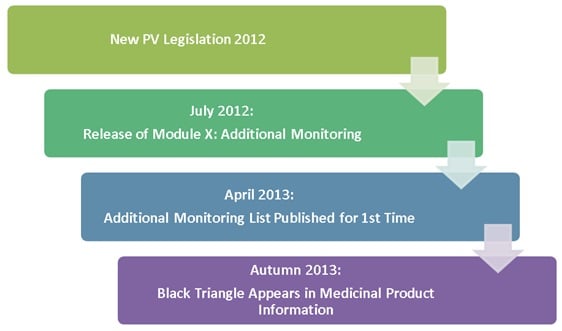
In July 2012 the new Pharmacovigilance legislation came into effect with a focus on robust systems for public health that encourage efficiency and transparency. The EMA Executive Director Guido Rasi explained that:
“The new pharmacovigilance legislation will help us to make the system more robust for public health and more transparent. It gives regulators a range of new or improved tools to ensure that patients are not exposed to unnecessary risks when taking medicines. It also increases the efficiency of medicines regulation for the benefit of all stakeholders.”
One of the new schemes within the new legislation is the adoption of the Black Triangle symbol, which becomes the EU’s symbol to strengthen surveillance and monitoring of medicines in its member states. This is not a new symbol nor is it a new scheme as it originated in the United Kingdom. For many organisations and individuals whose medicines are marketed in the United Kingdom, this can be seen as an extension of an existing process. It has been a familiar symbol on medicinal products for a number of years. What this does is strengthen a “tried and tested” process right across the EU.
▼ "This medicinal product is subject to additional monitoring"
From this autumn, the Black Triangle Symbol will begin to appear on package leaflets in medicinal products across EU member states. The inverted black triangle and statement above will be present as an indication of additional monitoring. It is important to note that the symbol will not appear on the outer packaging or labelling.
The aim is to encourage patients and healthcare professionals to report all suspected adverse reactions for medicines with the black triangle symbol. The inclusion of the Black Triangle allows patients and healthcare professionals to identify medicines that are being additionally monitored. The result it is hoped will be an increase in the number of suspected adverse reactions reported by patients and healthcare professionals, which will allow medicines to be monitored and analysed more efficiently in the early years of marketing authorisation.
In preparation for this in April 2013, the EMA released the Additional Monitoring List. This is published monthly on the EMA website with public access to the list of all medicines currently being monitored. Addition and removal of medicines from the list are the sole responsibility of the Pharmacovigilance Risk Assessment Committee (PRAC) who perform a monthly review before updates are published.
Which medicines will I find on the Additional Monitoring List?
• Any medicine that contains a new active substance authorised in the EU after 1 January 2011
• Any biological medicine, such as a vaccine or a medicine derived from plasma (blood), authorised in the EU after 1 January 2011
• Any medicine that has been given a conditional approval (where the company that markets the medicine must provide more data about it) or approved under exceptional circumstances (where there are specific reasons why the company cannot provide a comprehensive set of data);
• Any company that markets the medicine is required to carry out additional studies, for instance, to provide more data on long-term use of the medicine or on a rare side effect seen during clinical trials.
For more information, the EMA has released a short You Tube Video and a leaflet explaining the Black Triangle Symbol.


